What Is Chemotherapy-Induced Neuropathy?
Chemotherapy-induced peripheral neuropathy (CIPN) is an injury to peripheral nerves caused as a side effect of certain chemotherapeutic agents. It is one of the most frequent non-hematologic complications of cancer treatment, affecting up to 68% of patients in the first months of chemotherapy.
The most frequently associated agents include taxanes (paclitaxel, docetaxel), platinum compounds (cisplatin, oxaliplatin, carboplatin), vinca alkaloids (vincristine), bortezomib, and thalidomide. Each class causes neuropathy by distinct mechanisms.
Beyond the direct impact on quality of life (pain, loss of function, falls), CIPN can lead to dose reduction or premature chemotherapy discontinuation, potentially compromising cancer treatment efficacy. It's a significant clinical challenge because preventive and therapeutic options are limited.
High Incidence
Affects up to 68% of patients in the first months of chemotherapy. May persist for months or years after treatment ends.
Varied Mechanisms
Each class of chemotherapeutic agent damages nerves by different mechanisms — mitochondrial damage, microtubule disruption, or dorsal root ganglion injury.
Impact on Treatment
May require dose reduction or chemotherapy interruption, potentially compromising the oncologic outcome.
Pathophysiology
Mechanisms of neurotoxicity vary by chemotherapy class but converge on damage to sensory neurons of the dorsal root ganglion (DRG), peripheral axons, or both. The DRG is particularly vulnerable because it lies outside the blood-brain barrier.

MECHANISMS OF NEUROTOXICITY BY CLASS
| AGENT | MAIN MECHANISM | CLINICAL PATTERN |
|---|---|---|
| Taxanes (paclitaxel) | Excessive microtubule stabilization, blocking axonal transport | Distal sensory neuropathy, acute pain (paclitaxel acute pain syndrome) |
| Platinums (cisplatin, oxaliplatin) | DNA and mitochondrial damage to dorsal ganglion neurons | Sensory neuronopathy, "coasting" phenomenon after discontinuation |
| Vinca alkaloids (vincristine) | Microtubule depolymerization, axonal transport disruption | Sensorimotor neuropathy, constipation (autonomic) |
| Bortezomib | Proteasome inhibition, accumulation of toxic proteins in nerves | Painful small-fiber sensory neuropathy |
| Thalidomide | Damage to dorsal ganglion and inhibition of vasa nervorum angiogenesis | Cumulative axonal sensory neuropathy |
Symptoms
CIPN presents mainly as a symmetric distal sensory neuropathy with a "stocking-and-glove" pattern. Positive (pain, tingling) and negative (numbness, sensory loss) sensory symptoms coexist in varying degrees.
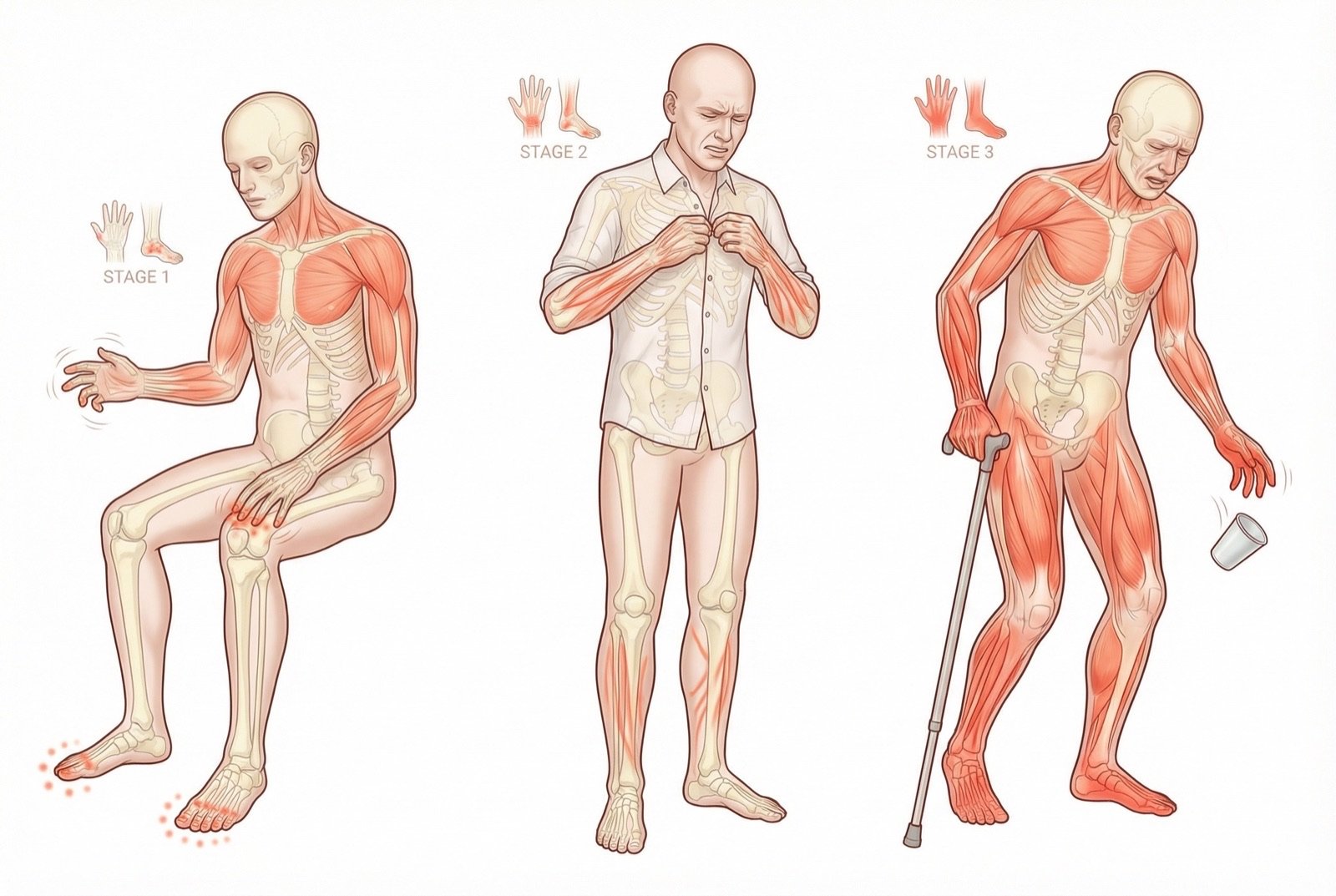
CIPN Symptoms
- 01
Tingling and paresthesias in hands and feet
Typically symmetric, starting at the fingertips. Often the first symptom patients notice.
- 02
Numbness and loss of sensation
Stocking-and-glove pattern progression. Sensory loss can impair buttoning, writing, or handling objects.
- 03
Neuropathic pain (burning, stabbing)
Burning, shock-like, or stabbing pain, often worse at night. May be the most limiting symptom.
- 04
Cold hypersensitivity (oxaliplatin)
Extreme cold sensitivity, including pain when touching cold objects or drinking cold liquids. Specific to oxaliplatin.
- 05
Loss of manual dexterity
Difficulty with fine motor tasks — buttoning, zipping, typing. Significant functional impact.
- 06
Postural instability and falls
Loss of foot proprioception, increasing risk of imbalance and falls, especially in older adults.
- 07
Muscle weakness (less frequent)
Most common with vincristine. Distal weakness in the feet (foot drop) and hands (grip weakness).
Diagnosis
CIPN diagnosis is clinical, based on the temporal relationship between starting neurotoxic chemotherapy and onset of neuropathic symptoms. A baseline neurologic assessment before chemotherapy is essential to identify preexisting neuropathy.
Severity is graded by the CTCAE (Common Terminology Criteria for Adverse Events) scale in grades 1 through 4, which guide decisions about dose adjustment. Accurate grading is fundamental to balancing oncologic control and quality of life.
🏥CIPN Grading (CTCAE v5.0)
- 1.Grade 1: Asymptomatic, diagnosed only on clinical examination. No intervention required.
- 2.Grade 2: Moderate symptoms, limiting instrumental activities (cooking, using a computer). Consider dose modification.
- 3.Grade 3: Severe symptoms, limiting self-care activities (dressing, eating). Discontinuation or significant dose reduction indicated.
- 4.Grade 4: Potentially life-threatening consequences. Immediate discontinuation of the agent indicated.
Differential Diagnosis
Not every neuropathy in a cancer patient is caused by chemotherapy. Coexisting or alternative causes must be actively ruled out, since some are treatable and can worsen the picture.
DIAGNÓSTICO DIFERENCIAL
Differential Diagnosis
Diabetic Neuropathy
Read more →- Preexisting T2DM
- Feet most affected
- Slow progression
Testes Diagnósticos
- Glycated hemoglobin
- EMG
Guillain-Barre Syndrome
- Acute/subacute onset
- Rapid progression
- Areflexia
- Progression over days = urgent admission
Testes Diagnósticos
- Lumbar puncture
- EMG
Nerve Root Compression by Metastasis
- Radicular pain in cancer patient
- May be unilateral
- Progression
- Progressive neurologic deficit in a cancer patient = emergency
Testes Diagnósticos
- Spine MRI
B12 Deficiency (chemotherapy-related)
- Symmetric paresthesias
- Megaloblastic anemia
- Smooth tongue
Testes Diagnósticos
- Serum vitamin B12
- Complete blood count
Paraneoplastic Neuropathy
- Associated with anti-Hu, anti-Yo antibodies
- May precede cancer diagnosis
- Sensory predominant
Testes Diagnósticos
- Paraneoplastic antibodies
- PET-CT
CIPN vs. Preexisting Diabetic Neuropathy
Diabetic patients starting chemotherapy are a special risk group: preexisting diabetic neuropathy increases nerve vulnerability to neurotoxic agents. Distinguishing each cause's contribution is essential for management. Key clues are the temporal relationship with chemotherapy initiation, abrupt worsening, and typical distribution of complaints (symmetric, distal, "stocking-and-glove"). Glycated hemoglobin and electroneuromyography (EMG) with nerve conduction studies help quantify the diabetic component.
Patients on neurotoxic agents whose neuropathy progresses faster than expected, with a prominent motor component, or with asymmetric deficit should raise suspicion of an alternative or coexisting cause — particularly Guillain-Barre syndrome, metastatic compression, or paraneoplastic neuropathy.
Paraneoplastic Neuropathy: Critical Differential Diagnosis
Paraneoplastic neuropathy can precede the cancer diagnosis by months or years and may be wrongly attributed to chemotherapy in patients already in treatment. It is characterized by severe sensory predominance, dorsal ganglion involvement (sensory neuronopathy), subacute progression, and often the presence of onconeural antibodies (anti-Hu, anti-Yo). PET-CT investigation can reveal an occult neoplasm.
Vitamin B12 deficiency is often overlooked in cancer patients. Beyond the effect of some chemotherapeutic agents on B12 absorption, the cancer itself and its treatments can compromise nutritional status. Serum B12 measurement should be part of the workup for any neuropathy in a cancer patient.
When to Suspect Spinal Cord Compression by Metastasis
In a cancer patient with new-onset neuropathy, vertebral metastasis with nerve root compression is a possibility that should never be dismissed without proper evaluation. Red flags include: recent-onset radicular pain, marked asymmetry, progression over days to weeks, urinary or bowel symptoms, and pain worsening when lying down. Contrast-enhanced spine MRI is the test of choice and must be ordered urgently when these signs are present.
The distinction between CIPN and metastatic compression has critical therapeutic implications: spinal cord compression is an oncologic emergency requiring urgent radiotherapy, high-dose corticosteroids, and possible surgical intervention. Any delay in diagnosis can result in permanent neurologic deficit.
Treatment
Managing CIPN is one of the greatest challenges in neuro-oncology. Proven therapeutic options are limited, and prevention (through monitoring and dose adjustment) remains the most important strategy.
Prevention
The American Society of Clinical Oncology (ASCO) does not recommend any neuroprotective agent for routine prevention of CIPN. Vitamins, antioxidants, glutamine, and calcium/magnesium have not demonstrated consistent efficacy in randomized trials. Effective prevention consists of rigorous clinical monitoring and dose adjustment when signs of neuropathy emerge.
Treatment of Neuropathic Pain
Duloxetine (60 mg/day) is the only agent with consistent clinical evidence for treatment of pain in established CIPN (Smith et al., JAMA 2013 — CALGB 170601; NNT ~6). The ASCO 2020 recommendation (Loprinzi et al., JCO) is moderate. ASCO 2020 explicitly advises against routine use of gabapentin (Rao 2007 — negative trial); amitriptyline also had a specific negative trial (Kautio 2008). Pregabalin may be tried case by case, with evidence extrapolated from diabetic neuropathy.
Non-pharmacologic approaches include physical exercise (growing evidence of benefit), physical therapy, occupational therapy (for functional compensation strategies), and psychological interventions (cognitive behavioral therapy for pain management).
Acupuncture as Treatment
Acupuncture has gained growing attention as a complementary therapy for CIPN, partly because pharmacologic options are limited. Several leading cancer centers already incorporate acupuncture into their integrative medicine services for CIPN patients.
The most relevant studies specifically for CIPN include Bao et al. (Journal of the National Cancer Institute, 2020, n=75) and Molassiotis et al. (Cancer, 2019) — randomized trials showing significant improvement in neuropathy symptoms with acupuncture compared with controls (waitlist/sham). Proposed mechanisms include improved microcirculation in peripheral nerves, modulation of nociceptive transmission in the dorsal horn, and release of neurotrophic factors.
In cancer patients, acupuncture is generally well tolerated when performed by an experienced physician acupuncturist, with prior assessment of platelet and neutrophil counts. Like any procedure, it carries risks (bleeding, infection) that must be weighed case by case. It can be particularly useful in patients with partial response to duloxetine or who don't tolerate the medication. Typical protocols involve weekly sessions for 8-12 weeks.
Prognosis
CIPN prognosis depends on the causative agent, cumulative dose, and severity at discontinuation. In general, taxane-induced neuropathy tends to improve gradually in the months after chemotherapy ends, although some patients retain residual symptoms.
Neuropathy from platinum compounds (especially cisplatin) has a more guarded prognosis, with the "coasting" phenomenon and slower, often incomplete recovery. Long-term follow-up studies show that 30% of cancer survivors still have clinically significant neuropathy years after treatment.
The impact on cancer survivors' quality of life is substantial — persistent CIPN affects mobility, sleep, manual function, and daily activities. Recognizing CIPN as a condition requiring chronic management is essential to comprehensive care of cancer survivors.
Myths and Facts
Myth vs. Fact
Neuropathy always improves when chemotherapy ends
Although many patients improve, up to 30% have significant symptoms months to years after treatment. With cisplatin, neuropathy can even worsen after discontinuation (coasting).
Vitamins and supplements prevent CIPN
No supplement has shown efficacy for preventing CIPN in robust clinical trials. ASCO does not recommend neuroprotective agents for routine prevention.
There is nothing to do to treat the pain
Duloxetine has robust evidence of efficacy for neuropathic pain in CIPN. Exercise, physical therapy, and acupuncture are complementary options with growing evidence.
Foot symptoms during chemotherapy don't need to be reported
Reporting symptoms early to the oncologist allows dose adjustments that can prevent permanent nerve damage without compromising treatment outcome.
When to Seek Help
Frequently Asked Questions
Frequently Asked Questions
Not necessarily. With taxanes, there is a tendency toward gradual improvement in the following months. With platinum compounds (cisplatin, oxaliplatin), neuropathy may continue worsening for weeks to months after discontinuation — a phenomenon called "coasting." Follow-up studies show that about 30% of cancer survivors maintain clinically significant neuropathy years after treatment. Recovery depends on the agent used, cumulative dose, and severity at discontinuation.
Yes, always. Reporting neuropathic symptoms early to the oncologist is the most important action a patient can take. Early detection allows dose adjustments or protocol changes that can prevent permanent nerve damage. There is no reason to "tough it out" in silence — the oncologist needs this information to make informed therapeutic decisions.
Current evidence does not support routine use of any supplement for CIPN prevention. ASCO has reviewed the data extensively and does not recommend vitamin E, glutamine, calcium/magnesium, acetyl-L-carnitine, or other agents for prevention. Some supplements, such as acetyl-L-carnitine, have shown neutral or even negative results in controlled studies. The most effective prevention is rigorous clinical monitoring with dose adjustment when needed.
Randomized trials specifically in CIPN (Bao et al., JNCI 2020; Molassiotis et al., Cancer 2019) showed improvement in neuropathy symptoms with acupuncture in patients with persistent CIPN, although the studies are modest in size. Proposed mechanisms include improved microcirculation in peripheral nerves, modulation of nociceptive transmission, and release of neurotrophic factors. The Society for Integrative Oncology and ASCO recognize acupuncture as a complementary option for symptom management in oncology. It should be performed by a physician acupuncturist experienced in integrative oncology.
The most neurotoxic are platinum compounds (cisplatin > oxaliplatin > carboplatin), taxanes (paclitaxel > docetaxel), vincristine, bortezomib, and thalidomide/lenalidomide. Cisplatin has the highest cumulative neurotoxicity and the worst recovery prognosis. Paclitaxel causes a characteristic acute pain syndrome in the days after infusion, on top of chronic neuropathy.
It's a valid concern and one of the main clinical dilemmas in neuro-oncology. Timely dose reduction — done before severe damage is established — generally does not significantly compromise the oncologic outcome in most protocols. Premature interruption due to severe neuropathy, however, can have an impact. That's why proactive monitoring and early dose adjustment (grades 2-3) are essential to balancing oncologic efficacy and neurologic preservation.
The most common pattern is a symmetric distal "stocking-and-glove" neuropathy, with predominance in feet and hands. However, in severe cases, it may progress proximally. Vincristine can cause autonomic neuropathy (constipation, urinary retention). High-dose cisplatin can affect hearing (ototoxicity). Some patients develop distal muscle weakness, especially with vincristine.
Coasting is the phenomenon where neuropathy from platinum compounds (especially cisplatin) keeps worsening for weeks to months after chemotherapy is discontinued. It happens because the primary damage is to neuronal cell bodies in the dorsal root ganglion, and axonal degeneration progresses even after the toxic agent is removed. It differs from taxane-induced neuropathy, which tends to stabilize and improve after discontinuation. Knowing about this phenomenon is crucial for therapeutic planning and recovery expectations.
Yes, significantly. Loss of foot proprioception (position sense) impairs balance and postural stability. Studies show a 3-5 times higher fall risk in patients with significant CIPN. Preventive measures include physical therapy with balance training, appropriate non-slip footwear, removing throw rugs and floor objects at home, installing grab bars in the bathroom, and adequate nighttime lighting.
It depends on the grade of neuropathy and the type of work. Mild neuropathy (grade 1-2) generally allows continued activity with adaptations. Loss of manual dexterity can impact occupations requiring fine skills. Postural instability can be a problem in jobs requiring balance or equipment operation. Occupational therapy can help with compensation strategies and adapting the work environment to maintain function.
Related Reading
Deepen your knowledge with related articles