What Is Diabetic Neuropathy?
Diabetic neuropathy is an injury to the peripheral nerves caused by chronic exposure to elevated blood glucose levels. It is the most common chronic complication of diabetes mellitus, affecting up to 50% of diabetic patients over their lifetime. It can involve sensory, motor, and autonomic nerves.
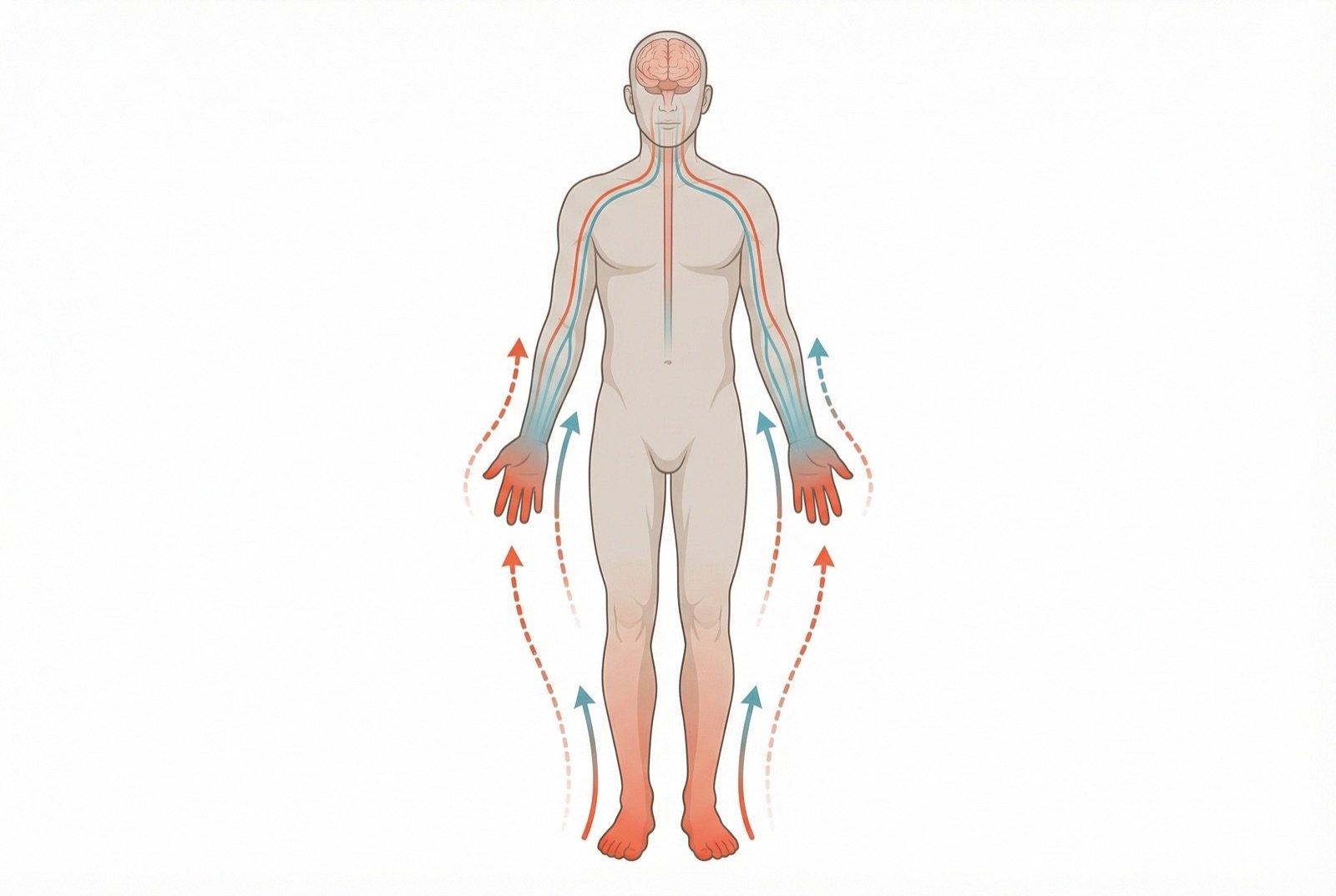
The most frequent form is distal symmetric polyneuropathy, which predominantly affects the feet and legs in a "stocking-and-glove" pattern, progressing in an ascending fashion. Symptoms range from mild tingling and numbness to intense, disabling neuropathic pain.
Beyond pain and discomfort, diabetic neuropathy is the leading cause of foot ulcers and non-traumatic amputations. Loss of protective foot sensation lets small injuries go unnoticed, progressing to ulcers and serious infections. Early diagnosis and treatment are essential to prevent these devastating complications.
High Prevalence
Affects up to 50% of diabetic patients. Can be present at type 2 diabetes diagnosis and even in prediabetes.
Loss of Protection
Loss of foot sensation is the leading cause of diabetic ulcers and non-traumatic amputations — a preventable complication.
Metabolic Mechanism
Chronic hyperglycemia causes direct nerve damage through oxidative stress, sorbitol accumulation, protein glycation, and microvascular damage.
Pathophysiology
Diabetic neuropathy results from a complex interaction between metabolic factors and vascular factors, both triggered by chronic hyperglycemia. These mechanisms lead to progressive damage of axons and Schwann cells (myelin-producing cells), affecting the longest nerve fibers first.

Metabolic Mechanisms
Polyol pathway: hyperglycemia increases the activity of the enzyme aldose reductase, which converts glucose to sorbitol. Sorbitol accumulation causes osmotic edema, depletion of myoinositol, and reduction of glutathione (an antioxidant), generating oxidative stress and cell damage.
Advanced glycation end products (AGEs): glucose binds non-enzymatically to structural nerve proteins, altering their function. AGEs also activate receptors (RAGE) that trigger inflammatory and oxidative-stress cascades, damaging axons and Schwann cells.
Oxidative stress: hyperglycemia increases free-radical production in mitochondria, exceeding the antioxidant capacity of nerve cells. This is considered the central unifying mechanism of diabetic neuropathic damage, connecting all metabolic pathways.
Vascular Mechanism
Peripheral nerves receive blood supply from small vessels (vasa nervorum). Hyperglycemia causes microangiopathy of the vasa nervorum — basement membrane thickening, lumen reduction, and decreased blood flow. The resulting chronic ischemia significantly contributes to nerve degeneration.
Symptoms
Diabetic neuropathy can manifest in several ways, depending on which nerve fibers are affected. The most common presentation is distal symmetric polyneuropathy with predominantly sensory symptoms.
Symptoms of Diabetic Neuropathy
- 01
Tingling and numbness in the feet
Distal paresthesias that typically start in the toes and progress upward. The first symptom most patients notice.
- 02
Burning or scorching pain
Burning neuropathic pain, often worse at night and at rest. Can be intense and disrupt sleep.
- 03
Spontaneous electric shocks
Sudden painful discharges with no apparent stimulus, reflecting ectopic activity in damaged nerves.
- 04
Allodynia
Pain on light touch — bedsheet contact with the feet at night can be unbearable.
- 05
Loss of sensation
Progressive loss of tactile, thermal, and pain sensation. The patient may not notice foot injuries.
- 06
Loss of balance
Impaired foot proprioception (position sense), increasing fall risk.
- 07
Distal muscle weakness
In advanced stages, intrinsic foot muscle weakness may occur, with deformities (claw toes).
- 08
Autonomic symptoms
Orthostatic hypotension, gastroparesis, erectile dysfunction, foot anhidrosis (dry, cracked skin).
CLINICAL FORMS OF DIABETIC NEUROPATHY
| FORM | FEATURES | PREVALENCE |
|---|---|---|
| Distal symmetric polyneuropathy | Stocking-and-glove pattern, symmetric, ascending | Most common (75%) |
| Autonomic neuropathy | Cardiovascular, gastrointestinal, genitourinary | Common (up to 50%) |
| Focal/multifocal neuropathy | Cranial mononeuropathy, truncal radiculopathy | Less common |
| Diabetic amyotrophy | Pain and proximal weakness in lower limbs | Rare (< 1%) |
Diagnosis
Diagnosing diabetic neuropathy combines clinical history, focused neurologic examination, and, when needed, complementary tests. Annual screening is recommended for all type 2 diabetes patients from diagnosis and for type 1 diabetes patients after 5 years of disease.
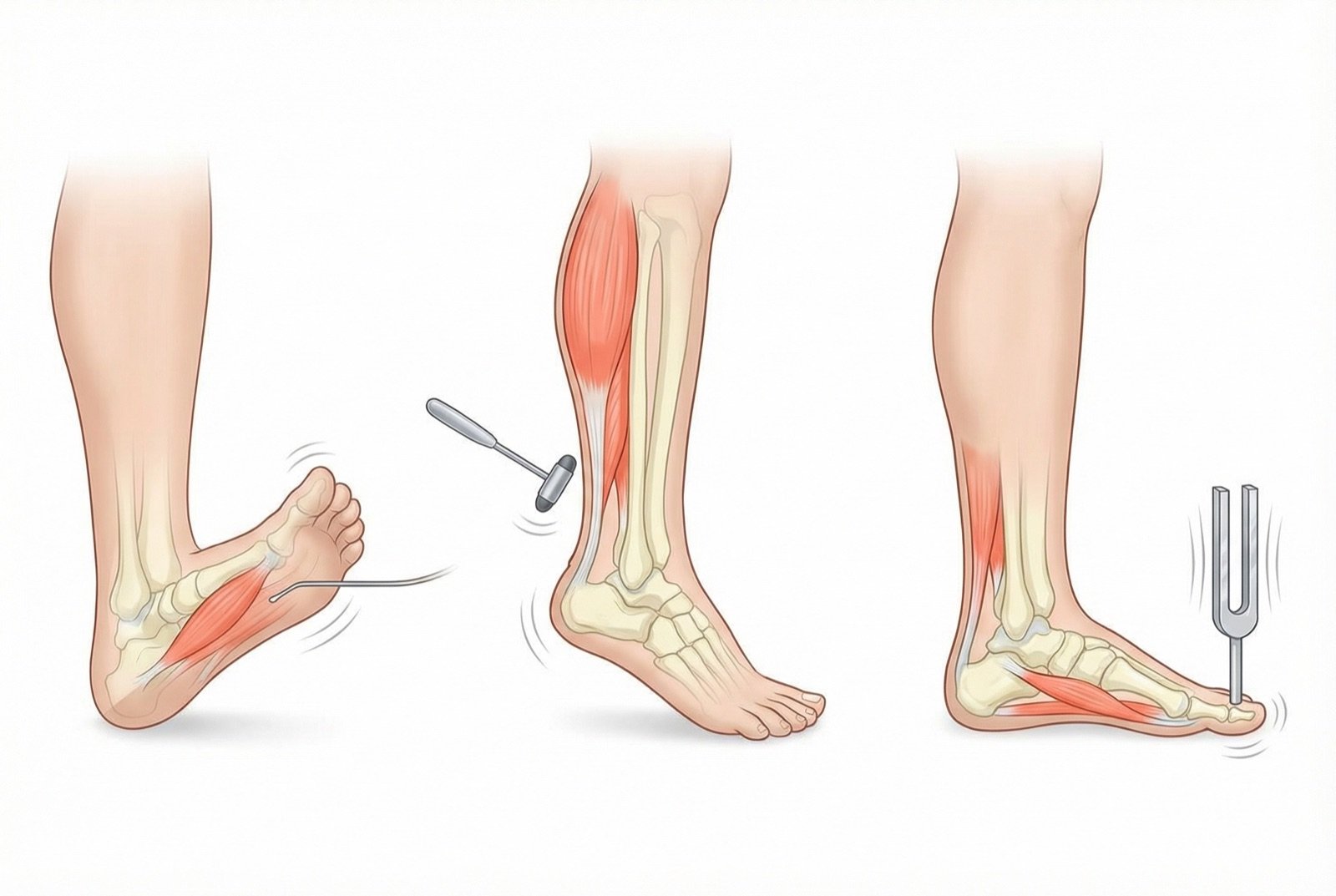
🏥Foot Neurologic Examination — Screening
- 1.10 g monofilament test (Semmes-Weinstein): assesses protective sensation. Loss identifies ulceration risk.
- 2.Vibration sensation test (128 Hz tuning fork): applied at the great toe. Assesses large myelinated fibers.
- 3.Thermal sensation test: ability to distinguish cold from warm at the feet.
- 4.Achilles reflexes: often diminished or absent early on.
- 5.Pinprick sensation test: assesses small nociceptive fibers.
Differential Diagnosis
Not every neuropathy in a diabetic patient is caused by diabetes. Treatable causes of neuropathy must be actively excluded, since they may coexist with diabetic neuropathy or be the actual cause of symptoms.
DIAGNÓSTICO DIFERENCIAL
Differential Diagnosis
Carpal Tunnel Syndrome
Read more →- Numbness limited to the median territory
- Positive Phalen sign
- Worse at night
Testes Diagnósticos
- EMG
Alcoholic Neuropathy
- Pattern similar to DN: distal, symmetric
- Chronic alcohol use
- Vitamin B1 deficiency
Testes Diagnósticos
- B1/B12 levels
- GGT
- EMG
Hypothyroidism
- Paresthesias + fatigue + weight gain
- Slow reflexes
- Elevated TSH
Testes Diagnósticos
- TSH
- Free T4
Chronic Inflammatory Demyelinating Polyradiculoneuropathy (CIDP)
- Predominant weakness
- Progression over more than 2 months
- CSF with albuminocytologic dissociation
- Progressive weakness = urgent neurologic evaluation
Testes Diagnósticos
- Nerve conduction velocity
- Lumbar puncture
Vitamin B12 Deficiency
- Neuropathy + megaloblastic anemia
- Glossitis
- Vegetarians/older adults
Testes Diagnósticos
- Serum B12
- Complete blood count
Diabetic Neuropathy vs Other Metabolic Neuropathies
Diabetic neuropathy is the most common cause of peripheral neuropathy in Western countries, but it often coexists with other treatable causes. Vitamin B12 deficiency is particularly relevant because it affects up to 30% of diabetic patients on chronic metformin — the drug reduces intestinal B12 absorption in a dose-dependent manner. The clinical picture can mimic diabetic neuropathy, and B12 deficiency often improves with adequate replacement, though the degree of recovery depends on the duration and severity of the condition.
Hypothyroidism is another metabolic cause that often overlaps — it can cause or worsen peripheral neuropathy and is more prevalent in diabetic patients (especially women). Routine TSH screening in every diabetic patient with neuropathy is advisable. Levothyroxine treatment can significantly improve neuropathic symptoms when hypothyroidism is the cause or an identified contributing factor.
When to Suspect Non-Diabetic Neuropathy
Certain patterns should raise suspicion that a diabetic patient's neuropathy has an additional or alternative cause. Warning signs include rapid progression (weeks to a few months), prominent muscle weakness, predominantly motor neuropathy, marked asymmetry, cranial nerve involvement, or neuropathy that does not match the degree of glycemic control.
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is more common in diabetics than in the general population and can present similarly to diabetic neuropathy, but with more prominent weakness and characteristically reduced nerve conduction velocity. Correct diagnosis is fundamental because CIDP responds to IV immunoglobulin, plasmapheresis, and corticosteroids — treatments that have no role in diabetic neuropathy.
Alcoholic Neuropathy: The Frequent Overlap
Alcoholic neuropathy has a clinical pattern almost identical to diabetic neuropathy — distal, symmetric, preferentially affecting the feet. Coexisting diabetes and alcoholism (not uncommon) makes clinical distinction even harder. Elements pointing to an alcoholic component include documented thiamine (vitamin B1) deficiency, history of significant alcohol consumption, elevated GGT, and partial improvement with abstinence and vitamin replacement.
The minimum laboratory work-up in every patient with peripheral neuropathy should include glucose/HbA1c, TSH, B12, folate, kidney function, complete blood count, and serum protein electrophoresis. This evaluation identifies the most common treatable causes and should precede the automatic attribution of every neuropathy to diabetes in diabetic patients.
Treatment
Diabetic neuropathy treatment involves three fundamental pillars: optimized glycemic control (the intervention with the greatest impact in preventing progression), treatment of neuropathic pain, and foot care.
Glycemic Control
Intensive glycemic control is the intervention with the most consistent evidence to prevent and slow the progression of diabetic neuropathy. In type 1 diabetes, the DCCT trial demonstrated a 60% risk reduction for neuropathy with intensive control. In type 2 diabetes, the benefit also exists, although less pronounced.
The glycated hemoglobin (HbA1c) target should be individualized but generally below 7%. Very rapid glucose reductions in patients with chronic poor control can paradoxically cause treatment-induced neuropathy of diabetes (transient acute neuropathic pain).
Treatment of Neuropathic Pain
Diabetic neuropathic pain does not respond to common analgesics. First-line drugs are: pregabalin (150-600 mg/day) and duloxetine (60-120 mg/day), both with strong evidence of efficacy. Gabapentin (900-3600 mg/day) and amitriptyline (25-75 mg/day) are second-line options.
Topical capsaicin 8% (patch) is indicated for localized pain. In refractory cases, tramadol or strong opioids may be necessary, but cautiously. Drug combinations across different classes (for example, pregabalin + duloxetine) may be more effective than monotherapy.
Foundation: Glycemic Control
HbA1c < 7% (individualized). The intervention with the most consistent evidence for preventing and slowing progression. Control associated cardiovascular risk factors.
1st-Line for Pain: Pregabalin or Duloxetine
Start at low dose and titrate gradually. Assess efficacy at 4-8 weeks. Pregabalin: 75 mg twice daily, up to 300 mg twice daily.
2nd-Line: Gabapentin or Amitriptyline
Indicated when first-line therapy is ineffective or not tolerated. Amitriptyline: use caution in older adults and patients with cardiac disease.
Adjunct: Topical Capsaicin and Combinations
Capsaicin 8% for localized pain. Drug combinations across different classes. Non-pharmacologic approaches.
Acupuncture as Treatment
Acupuncture has been studied as complementary therapy in peripheral diabetic neuropathy, with variable and still limited results in some clinical trials. Proposed mechanisms — predominantly from experimental studies — include possible effects on microcirculation, central pain modulation via the endogenous opioid pathway, and influence on oxidative-stress markers; clinical translation of these findings still requires more robust evidence.
Experimental studies in animal models of diabetic neuropathy show that acupuncture can improve nerve conduction velocity and reduce oxidative-stress markers in peripheral nerves. Human clinical trials suggest improved sensory symptoms and quality of life, though higher-quality studies are needed.
Acupuncture may be considered as part of a multimodal approach, especially in patients with partial response to pharmacologic treatment or who experience significant side effects. Glycemic control should always remain the foundation of treatment.
Prognosis
Diabetic neuropathy is a chronic and generally progressive condition. With adequate glycemic control, progression can be significantly slowed or stabilized. However, reversal of established nerve damage is limited, especially in advanced forms.
The most significant long-term impact is the risk of foot ulceration and amputation. Diabetic patients with neuropathy have an annual ulceration risk of 7% and an amputation risk 15-40 times greater than diabetics without neuropathy. Structured foot-care programs dramatically reduce these complications.
Neuropathic pain may improve over time in some patients (as nerve fibers degenerate completely), but this represents worsening neuropathy, not improvement. Progressive sensory loss is the most dangerous long-term scenario.
Myths and Facts
Myth vs. Fact
Tingling in the feet is normal with age
Persistent foot tingling is not normal at any age. In diabetic patients, it is a warning sign that should be investigated as possible neuropathy.
If it does not hurt, there is no neuropathy
Up to 50% of patients with diabetic neuropathy are asymptomatic. The painless form with sensory loss is actually the most dangerous, since it raises ulcer risk.
Neuropathy only occurs in advanced diabetes
It can be present at type 2 diabetes diagnosis (since hyperglycemia can precede diagnosis by years) and even in prediabetes.
Common analgesics resolve neuropathic pain
Neuropathic pain does not respond adequately to simple analgesics or anti-inflammatories. It requires specific drugs such as pregabalin, duloxetine, or gabapentin.
There is nothing to do — neuropathy only worsens
Adequate glycemic control can significantly prevent and slow progression. Pain treatments are effective. Foot care prevents serious complications.
When to Seek Help
Frequently Asked Questions
Frequently Asked Questions
In early phases, intensive glycemic control can lead to partial neuropathy improvement, especially of symptoms. However, established axonal damage is rarely reversed — the main goal of glycemic control is to prevent progression, not reverse damage already established. In type 1 diabetes, the DCCT trial showed a 60% reduction in neuropathy risk with intensive control. The window for effective prevention is in the early years of diabetes.
Diabetic neuropathic pain classically worsens at rest and at night through several mechanisms: absence of daytime sensory stimuli that "distract" the central nervous system, reduction in foot temperature during sleep (which can exacerbate the ectopic activity of damaged nerves), and possibly the circadian rhythm of inflammatory mediators. Nighttime worsening is a feature that distinguishes neuropathic pain from muscular or articular pain, which generally worsens with use.
Yes, the vast majority of diabetic foot ulcers and amputations are preventable. Essential measures include: daily foot inspection (especially between the toes), heel moisturizing with cream, appropriate footwear without internal seams (preferring shoes with a wide toe box), socks without constrictive elastic, regular podiatric consultation, and annual monofilament screening to detect loss of protective sensation. Patient education about foot care is the highest-impact intervention for preventing amputations.
Metformin itself does not cause neuropathy, but it reduces small-intestinal vitamin B12 absorption in a dose- and time-dependent manner. B12 deficiency can occur in up to 30% of chronic metformin users and can cause or worsen peripheral neuropathy. For this reason, periodic vitamin B12 measurement is recommended for patients on long-term metformin, especially above 1,500 mg/day. B12 replacement is simple and effective.
Acupuncture has been studied as a complement in treating diabetic neuropathic pain, with variable results in clinical trials and still limited evidence. Proposed mechanisms — mostly from experimental studies — include possible effects on microcirculation, central pain modulation via the endogenous opioid pathway, and influence on oxidative-stress markers. The acupuncture physician may consider acupuncture as an adjunct to pharmacologic treatment, especially in patients with partial response to pregabalin or duloxetine. Glycemic control remains the foundation of treatment.
Both are first-line with similar efficacy. The choice depends on patient profile: duloxetine is preferred in patients with concomitant depression (treats both), obesity (may cause mild weight loss), or those already taking other sedating medications. Pregabalin is more useful with associated insomnia (useful hypnotic effect), generalized anxiety, or when duloxetine was not tolerated. Combining the two may be more effective than monotherapy in refractory cases.
Loss of protective sensation in diabetic neuropathy tends to progress without adequate glycemic control. With optimized metabolic control, progression can be greatly slowed or stabilized. Significant recovery of large-fiber sensation is less common in adults with established neuropathy, though in type 1 diabetics with very good control there are data showing partial recovery. Small fibers (which mediate temperature and pain) may show more plasticity than large fibers.
Yes, diabetic autonomic neuropathy can have serious consequences. Cardiovascular autonomic neuropathy raises the risk of sudden death and silent myocardial infarction (painless, since cardiac nociceptive fibers are damaged). Orthostatic hypotension can cause falls and syncope. Gastroparesis impairs glycemic control. Erectile dysfunction affects quality of life. Neurogenic bladder raises urinary infection risk. Systematic screening for these complications is part of comprehensive diabetic care.
There is no absolute threshold — neuropathy risk increases continuously with duration of hyperglycemia exposure. The main predictor is mean glycated hemoglobin (HbA1c) over the years. Each 1% reduction in chronically elevated HbA1c is associated with a significant drop in neuropathic complication risk. Important: neuropathy can already be present in prediabetes (HbA1c 5.7-6.4%), when glucose does not yet meet diabetes criteria, underscoring the importance of early screening.
Yes, daily foot inspection is one of the most important and lowest-cost preventive measures. Patients should check for blisters, cuts, cracks, calluses, redness, localized warmth, deformities, or color changes. Patients with vision difficulty or reduced mobility should use a mirror or ask a family member for help. Any injury, no matter how small, should be reported to the physician immediately — in the insensate diabetic foot, ignored small injuries quickly evolve into deep ulcers.
Related Reading
Deepen your knowledge with related articles