What Is Pain?
The International Association for the Study of Pain (IASP), in its revised 2020 definition, defines pain as "an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage." This definition has two fundamental consequences for patients.
First: pain is always a personal experience — no one can measure your pain from the outside. Second: pain is simultaneously sensory and emotional. There is no purely "physical" pain detached from emotions, nor "psychological" pain that is not real. All pain is processed by the brain, and therefore all pain is real.
Understanding this is the first step toward treating pain effectively — especially when it persists for weeks, months, or years without an obvious structural cause.
Pain Is Produced by the Brain
Tissue sends warning signals, but it is the brain that decides — based on context, memory, and emotions — whether to produce pain and at what intensity.
Pain Has a Protective Function
Acute pain is a biological alarm essential for survival. People with congenital inability to feel pain suffer severe injuries and have a shorter life expectancy.
Pain without Damage Is Possible
The brain can generate intense pain without current tissue injury. Likewise, severe injuries can occur without pain (as in war or extreme trauma).
Nociceptors: The Threat Detectors
The body is covered by specialized nerve endings called nociceptors. They are present in skin, muscles, joints, viscera, and virtually all tissues — except the brain itself, which has no nociceptors (which is why brain surgery can be performed with the patient awake).
Nociceptors respond to potentially damaging stimuli: extreme temperature, intense pressure, and chemicals released by injured tissue (such as bradykinin, prostaglandins, and substance P). Once activated, they transmit electrical signals to the spinal cord through two main nerve fiber types:
The A-delta fibers are myelinated and conduct rapidly — they are responsible for the immediate, sharp, localized acute pain you feel when you step on a nail. The C fibers are unmyelinated and conduct slowly — they are responsible for the dull, diffuse, persistent pain that continues minutes after the same injury.
PAIN NERVE FIBERS: COMPARISON
| FEATURE | A-DELTA FIBERS | C FIBERS |
|---|---|---|
| Myelination | Yes (fast conduction) | No (slow conduction) |
| Speed | 5-30 m/s | 0.5-2 m/s |
| Type of pain | Sharp, localized, vivid | Dull, diffuse, persistent |
| Sensation | First impact of damage | Pain that "comes after" |
| Example | Needle prick | Post-exercise muscle pain |
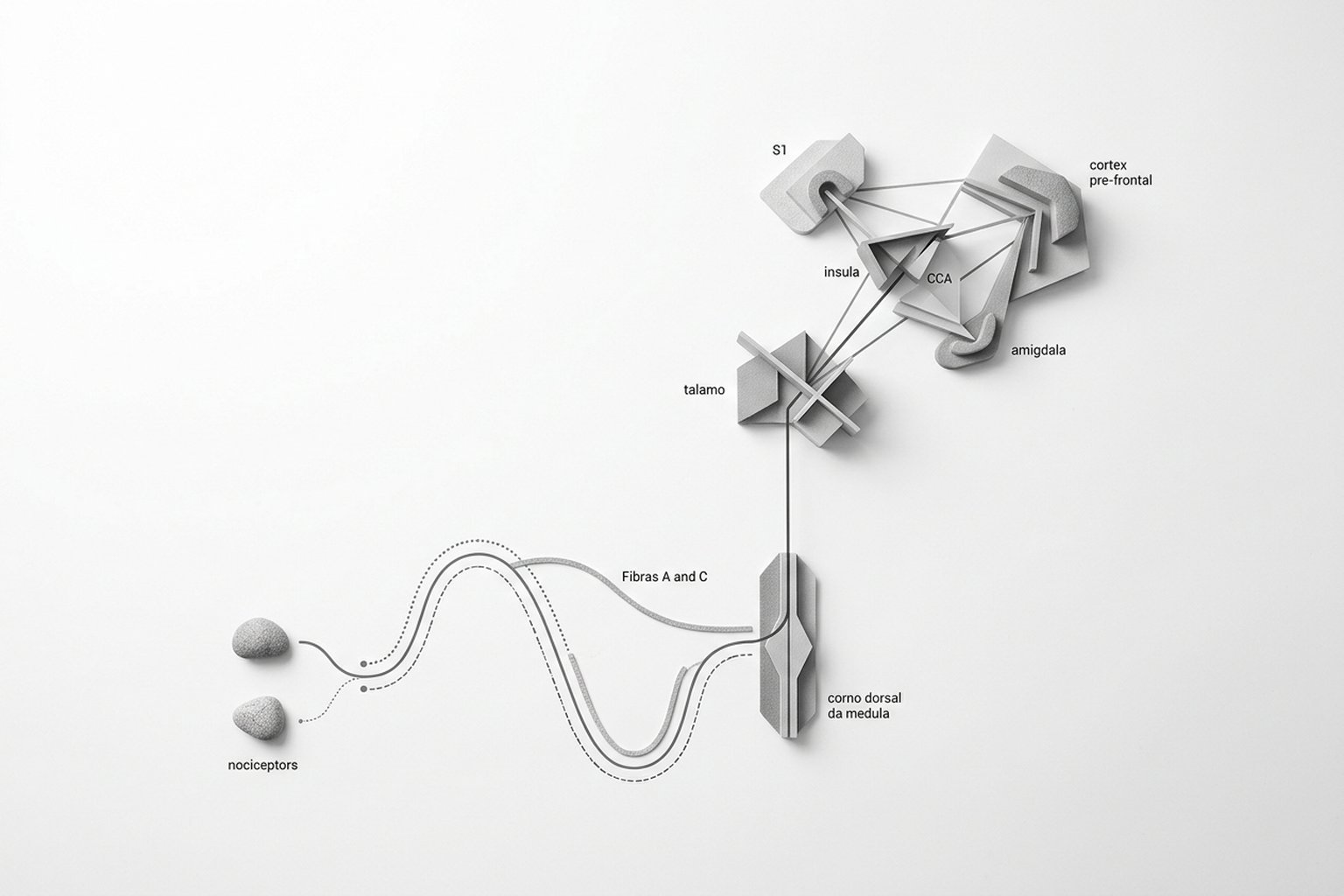
From the Spinal Cord to the Brain: The Path of the Signal
Signals from nociceptors reach the spinal cord through the dorsal root, where they synapse in the dorsal horn. Here the first processing occurs: the signal can be amplified (facilitation) or reduced (inhibition), depending on descending pathways from the brain and on local interneurons. That is why rubbing a sore área relieves pain — tactile fibers (A-beta) compete with nociceptive fibers in the dorsal horn.
From the spinal cord, the signal ascends through the spinothalamic tract to the thalamus, which distributes it to multiple brain áreas — the só-called "pain matrix":
Pain and Emotion: Shared Circuits
It is no coincidence that the language of physical and emotional pain blurs together: "broken heart," "heartache," "anguish." Neuroimaging studies (fMRI) show that social rejection and affective loss activate brain regions overlapping with physical pain (especially the anterior cingulate córtex and the insula) — although more refined analyses with MVPA also suggest distinct neural signatures between physical and social pain.
This has direct clinical implications: anxiety, depression, and chronic stress amplify pain perception because they share neural pathways. Treating chronic pain without addressing the patient's emotional state is like trying to put out a fire while ignoring half the flames.
Likewise, persistent pain alters mood and increases depression risk. This bidirectional relationship reinforces the need for a biopsychosocial approach.
"Pain is always real for the person who feels it. Doubting the patient's pain is the first barrier to effective treatment."
Why Does the Same Stimulus Hurt Differently in Each Person?
One of pain neuroscience's most fascinating findings is that the intensity of pain is not proportional to tissue injury. Soldiers wounded in battle frequently report little pain at the moment of injury — the brain, in survival mode, suppresses pain because it would interfere with escape. Conversely, an athlete who fears a serious injury may feel pain disproportionate to a minor wound.
What determines the intensity of pain is the brain's judgment about the level of threat the signal represents. This judgment is influenced by:
This explains why two patients with identical MRI images — showing the same disc herniation — can have completely different experiences: one with disabling pain, the other with none. Structural injury is just one factor among many.
Myths and Facts about Pain
Myth vs. Fact
If imaging studies are normal, the pain is psychological and not real.
Pain without structural findings on imaging is extremely common and biologically real. The nervous system can produce intense pain through central sensitization, dysregulation of descending inhibitory systems, and neural plasticity — without any visible lesion on imaging.
Myth vs. Fact
Pain means there is tissue damage — the more it hurts, the greater the damage.
Pain intensity does not reflect injury size. A small cavity can hurt immensely; an extensive muscle injury can hurt little. Pain is an alarm calibrated by multiple factors — not a damage gauge.
Myth vs. Fact
Chronic pain is inevitable — nothing can be done once it passes 3 months.
Chronic pain is treatable. The nervous system has plasticity — it can be 'reprogrammed' with the right approaches: pain education, medical acupuncture, graded exercise, sleep management, and psychological support. Many patients significantly reduce pain even after years of suffering.
When to Seek Medical Help
All pain deserves proper medical evaluation. Some signs indicate that specialized care is urgent.
Frequently Asked Questions about Pain Neuroscience
According to the International Association for the Study of Pain (IASP, 2020), pain is "an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage." This definition recognizes that pain is always a personal experience, is simultaneously sensory and emotional, and can exist even without current tissue injury.
Nociceptors are nerve endings specialized in detecting potentially damaging stimuli — extreme temperature, intense pressure, and chemicals released by injured tissue. They transmit electrical signals to the spinal cord through A-delta fibers (sharp, fast pain) and C fibers (dull, persistent pain). Importantly, nociceptors detect threats but do not produce pain — that decision belongs to the brain.
The brain produces pain in response to signals interpreted as a threat. It can generate intense pain without current tissue injury (as in phantom limb pain or fibromyalgia) because the central nervous system becomes hypersensitive. Conversely, the brain can suppress pain even with serious injury (as in extreme trauma). Pain is always real for the person who feels it, regardless of test findings.
Pain is processed by a network of brain áreas called the "pain matrix": the primary somatosensory córtex (where and how much it hurts), the insula (emotional and autonomic dimension), the anterior cingulate córtex (suffering and motivation to protect oneself), the amygdala (emotional memory of pain), and the prefrontal córtex (meaning and context). There is no single "pain center" — it is distributed.
Because anxiety, depression, and physical pain share brain circuits — especially the anterior cingulate córtex and the insula. Emotional state directly influences descending pain modulation systems, which can increase or reduce pain sensitivity. Stress also elevates HPA axis activity (cortisol) and the sympathetic nervous system, amplifying pain perception.
Yes. Conditions such as fibromyalgia, irritable bowel syndrome, chronic tension headache, and chronic low back pain frequently occur without identifiable structural injury on tests. In these cases, the central nervous system is in a state of hypersensitivity (central sensitization), generating amplified pain without proportional nociceptive input. This does not mean the pain is "imagination" — it is a real biological dysfunction of the nervous system.
The pain matrix is the distributed network of brain áreas that collectively produce the pain experience. It includes sensory regions (which localize and quantify pain), affective regions (which process associated suffering), cognitive regions (which assess meaning and context), and autonomic regions (which coordinate physical responses). Because pain involves this broad network, expectations, memories, and emotions só deeply modify the experience.
Medical acupuncture acts at multiple levels of the pain-processing system: locally, it activates A-delta fibers and releases adenosine (a local analgesic); in the spinal cord, it triggers enkephalin release and modulates nociceptive transmission; supraspinally, it activates the periaqueductal gray (PAG) and nucleus raphe magnus, which release endogenous opioids, serotonin, and norepinephrine. It also engages DNIC (Diffuse Noxious Inhibitory Controls; in updated IASP nomenclature: CPM — Conditioned Pain Modulation), diffuse nociceptive inhibitory controls that modulate pain transmission at the spinal level, reducing central hypersensitivity.
Partially. Imaging shows structural changes (herniations, osteoarthritis, inflammation), but the correlation between findings and pain is weak. Studies show that 30-40% of asymptomatic adults have visible disc herniations on MRI. Imaging does not measure nervous system sensitivity, emotional state, or psychosocial factors — which are often the main determinants of chronic pain.
Patients who understand the mechanisms of pain — that the brain produces it as a modifiable alarm, that pain does not necessarily equal damage, and that factors such as sleep, emotions, and movement influence pain — respond better to treatment. Studies show that "pain neuroscience education" reduces catastrophizing, improves adherence to movement, and increases the effectiveness of interventions. That is why medical consultations that explain these concepts have real therapeutic value.
Related Reading
Deepen your knowledge with related articles